Launched this week

Storke
AI co-pilot for medical device regulation, in Microsoft Word
2 followers
AI co-pilot for medical device regulation, in Microsoft Word
2 followers
Storke is an AI regulatory co-pilot inside Microsoft Word. Our knowledge engine covers EU MDR, UK MDR, FDA, ISO 13485, ISO 14971, and all major MEDDEV and MDCG guidances, with more global jurisdictions being added. It writes clinical evaluation plans, conducts literature reviews, generates GSPR checklists, and reviews documents for compliance inside Word. What takes consultants days, Storke does in minutes. Live with paying customers and piloting with consultancies across Europe.

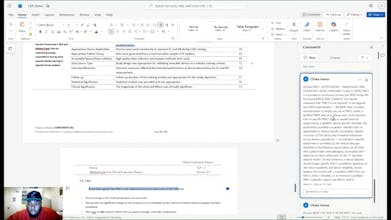

Hey Product Hunt!
I'm Chuka, co-founder of Storke. We're a multidisciplinary team with backgrounds in medicine, mathematics and political economy from UCL, Imperial, King's College London and Cambridge; additional backgrounds in data/ops @TikTok, AI policy @ UK Parliament, biotech simulation engineering and AI engineering for edtech.
We built this because we lived the problem of medical device regulations ourself. Our previous startup needed a medical device approved and the regulatory costs were absurd. So we built a knowledge engine from the ground up that understands how regulatory frameworks connect across jurisdictions at a depth we haven't seen anywhere else, including in published scientific research from institutions and some of our largest competitors.
...Then we put it inside Microsoft Word, where the work actually happens.
Every medical device that reaches a patient starts with a stack of regulatory documents. We're making sure that process never holds back innovation. Would love your feedback.